Paolo Lamagni, Matteo Miola, Jacopo Catalano, Mathias S Hvid, Mohammad Aref H Mamakhel, Mogens Christensen, Monica R Madsen, Henrik S Jeppesen, Xin‐Ming Hu, Kim Daasbjerg, Troels Skrydstrup, Nina Lock
https://doi.org/10.1002/adfm.201910408
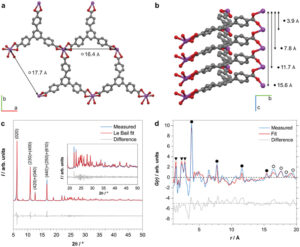
Recently, a large number of nanostructured metal‐containing materials have been developed for the electrochemical CO2 reduction reaction (eCO2RR). However, it remains a challenge to achieve high activity and selectivity with respect to the metal load due to the limited concentration of surface metal atoms. Here, it is reported that the bismuth‐based metal–organic framework Bi(1,3,5‐tris(4‐carboxyphenyl)benzene), herein denoted Bi(btb), works as a precatalyst and undergoes a structural rearrangement at reducing potentials to form highly active and selective catalytic Bi‐based nanoparticles dispersed in a porous organic matrix. The structural change is investigated by electron microscopy, X‐ray diffraction, total scattering, and spectroscopic techniques. Due to the periodic arrangement of Bi cations in highly porous Bi(btb), the in situ formed Bi nanoparticles are well‐dispersed and hence highly exposed for surface catalytic reactions. As a result, high selectivity over a broad potential range in the eCO2RR toward formate production with a Faradaic efficiency up to 95(3)% is achieved. Moreover, a large current density with respect to the Bi load, i.e., a mass activity, up to 261(13) A g−1 is achieved, thereby outperforming most other nanostructured Bi materials.