Jiaying Yu, Yunshuo Yang, Runtao Jia, Kim Daasbjerg, Jun Wang, Mauro Carraro, Zhuo Xin, Yuxing Huang, Troels Skrydstrup
https://doi.org/10.1016/j.apsusc.2020.147080
Large-scale synthesis of metal/carbon hybrids with tunable metal/carbon heterointerfaces is vital for practical application of such hybrids in electrocatalysis. Herein, we developed a facile route for large-scale crafting of ultrafine Ru nanoparticles (NPs) anchored on the ketonic C O groups of carbon nanotubes (CNTs) (9.6 g in one batch). From both experimental results and theoretical calculations, we demonstrate that C
O groups of carbon nanotubes (CNTs) (9.6 g in one batch). From both experimental results and theoretical calculations, we demonstrate that C O (rather than C–O) groups can be exploited to engineer the heterointerface of CNTs and Ru NPs. In fact, the electronic structure of Ru becomes considerably improved because the high polarity of C
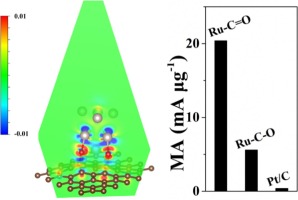
O (rather than C–O) groups can be exploited to engineer the heterointerface of CNTs and Ru NPs. In fact, the electronic structure of Ru becomes considerably improved because the high polarity of C O facilitates the interface electron transfer of Ru/CNTs. Consequently, the obtained Ru-O-CNTs hybrids with low Ru loading of 1.5 wt% display a small overpotential of 25 mV at 10 mA cm−2 and with fast kinetics as deduced from a small Tafel slope of 20.4 mV dec−1 for hydrogen evolution reaction (HER) in 1 M KOH. Impressively, at 70 mV overpotential, the hybrids exhibit a record mass activity of 20.4 mA μg−1Ru, which is more than 50-fold of that for commercial Pt/C. Therefore, for the first time, we identified the vital role of C
O facilitates the interface electron transfer of Ru/CNTs. Consequently, the obtained Ru-O-CNTs hybrids with low Ru loading of 1.5 wt% display a small overpotential of 25 mV at 10 mA cm−2 and with fast kinetics as deduced from a small Tafel slope of 20.4 mV dec−1 for hydrogen evolution reaction (HER) in 1 M KOH. Impressively, at 70 mV overpotential, the hybrids exhibit a record mass activity of 20.4 mA μg−1Ru, which is more than 50-fold of that for commercial Pt/C. Therefore, for the first time, we identified the vital role of C O groups for engineering metal/carbon heterointerfaces towards robust and efficient electrocatalysis.
O groups for engineering metal/carbon heterointerfaces towards robust and efficient electrocatalysis.