Line Koefoed, Steen U. Pedersen and Kim Daasbjerg
doi.org/10.1002/celc.201500512
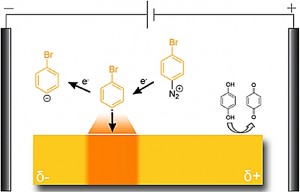
Bipolar electrochemistry makes various kinds of surface modifications possible by appropriately tuning the applied potential difference. In this study aryl diazonium, iodonium, and sulfonium salts are reductively grafted onto gold surfaces using an excess of hydroquinone to set the anode at a fixed potential. In general, aryl diazonium salts are easier to reduce than the corresponding iodonium salts which, again, are more easily reduced than sulfonium salts. For all three salts grafting is observed at the reductive site of the bipolar electrode, but with distinctly different features due to the different reduction potentials required to facilitate the grafting. Depending on the reduction potentials of the salts relative to that of the grafting agent, that is, the aryl radical, it is possible to create movable band-like grafted areas by diazonium grafting, expanding bands by iodonium grafting, or optically invisible thin bands by sulfonium grafting. From these findings, a relationship between the grafting behavior and the applied potential difference is deduced.